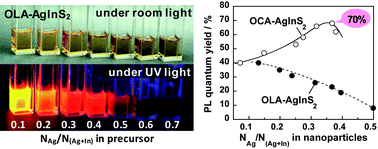
Non-stoichiometric AgInS2 (AIS) semiconductor particles were synthesized by the thermal decomposition of single-source precursors in solutions of two kinds of primary amines (oleylamine and octylamine). The Ag content in the resulting nanoparticles was controlled by adjusting the chemical composition of the precursor used, in which the mole ratio of Ag+ to total metal ions was varied from 0.1 to 0.7, resulting in the production of non-stoichiometric AIS particles with varying amounts of Ag vacancies. The average size of AIS particles was slightly decreased from 4.3 to 3.8 nm on changing the solvent from oleylamine to octylamine in the preparation, while the particle size seemed to be constant regardless of the content of Ag. On the other hand, the optical properties of AIS particles were considerably modified depending on the Ag content in the particles. The absorption onset was blue-shifted from 750 to 580 nm with a decrease in the Ag content, due to the enlargement of the energy gap of particles. Intense photoluminescence originating from the donor–acceptor pair recombination was observed for each kind of AIS particle and then the photoluminescence peak wavelength was also blue-shifted from 830 to 650 nm, being similar to the behavior of the absorption onset. The maximum photoluminescence quantum yield was ca. 70% for octylamine-modified AIS nanoparticles having a ratio of Ag+ to total metal ions of 0.37, which probably contained the optimum amount of Ag vacancies acting as the sites of donor–acceptor pair recombination with few surface defect sites for non-radiative recombination.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...