Bipolar cyano-substituted pyridine derivatives for applications in organic light-emitting devices†
Abstract
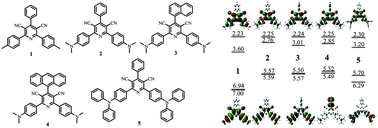
A novel series of cyano-substituted pyridine derivatives have been synthesized and characterized. Besides the expected electron-transporting (ET) properties stemming from the pyridine structure, the cyanopyridines also show light-emitting (EM) as well as bipolar transporting properties upon incorporation of larger π-conjugated groups, such as N,N-dimethylbenzenamine and triphenylamine groups. With these multifunctional cyanopyridines, device structures can be simplified to use only two layers or even a single layer. Low turn-on and driving voltages, relatively high power efficiency ηp and mild efficiency roll-off were demonstrated by the devices based on the compounds with better bipolar properties. The compound 2,6-bis(4-(diphenylamino)phenyl)-4-phenylpyridine-3,5-dicarbonitrile (PPPD) containing triphenylamine groups has relatively better hole-transporting (HT) ability. A device with a configuration of ITO/NPB (10nm)/PPPD (60nm)/BPhen (30nm)/LiF (0.5nm)/Mg: Ag (100nm) (where NPB is α-napthylphenylbiphenyl diamine, and BPhen is 7-diphenyl-1,10-phenanthroline), using PPPD as HT and EM layer, yields high efficiencies of 8.5 cd A−1 and 6.9 lm W−1 due to the balanced charge transporting. It is also found that substituting larger π-conjugated groups at the 4-position of the pyridine ring has a weak effect on the molecular properties because of the steric hindrance of the two cyano groups, suggesting a good linkage type for joining the cyano-substituted pyridine moiety with other HT moieties for further bipolar molecular design strategies.

 Please wait while we load your content...
Please wait while we load your content...