Thermodynamic and structural insight into the underlying mechanisms of the phosphatidylcholine liposomes – casein associates co-assembly and functionality†
Abstract
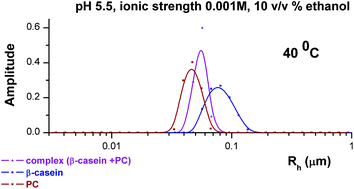
The combination of data obtained from isothermal mixing calorimetry and light scattering allowed us to reveal the relationships between the character of the interactions of casein (β-casein and sodium caseinate (SCN) particles) with phosphatidylcholine (PC) liposomes and their specific properties, such as, the hydrophilic–lipophilic balance of the surface properties, the size, and the architecture. The size distribution diagrams, obtained by dynamic light scattering for the particles of pure PC, β-casein and their complexes, indicated the involvement of both the protein and liposome particles in the complex formation at pH 5.5 and low ionic strength (0.001 M). As this took place, the data of the extraction of the free PC from the aqueous solutions of the complex particles by diethyl ether showed about 90% of the binding of the PC liposomes by the β-casein particles. As a result of the interactions, the complex particles showed markedly higher values of their density, calculated on the basis of the light scattering data, as compared to the ones, which were inherent to the pure protein particles. The generality of the importance of such a structural parameter as the density of the complex (protein + PC) particles for their function as protectors against oxidation for the unsaturated PC was supported by the data obtained for the complex particles formed between SCN and PC liposomes at pH 5.5, 40 °C and the low ionic strength (0.001 M). In addition, the importance of both the density and the architecture of the complex (protein + PC) particles for their susceptibility to the proteolysis in the gastrointestinal tract was illustrated by the example of the proteolysis of the complex (β-casein + PC) particles in the simulated gastrointestinal conditions in vitro.
- This article is part of the themed collection: Delivery of functionality in complex food systems

 Please wait while we load your content...
Please wait while we load your content...