Origin of the 3.6 V to 3.9 V voltage increase in the LiFeSO4F cathodes for Li-ion batteries†
Abstract
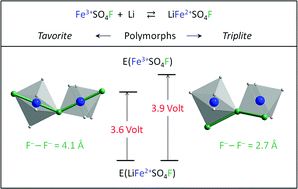
Recently, the LiFeSO4F material has been reported as the highest potential Fe-based cathode material for Li-ion batteries. Its working voltage vs. Li+/Li0 jumps from 3.6 V to 3.9 V when LiFeSO4F is synthesized with the fully ordered tavorite structure and the fully disordered triplite structure, respectively. The present study aims at rationalizing this voltage increase by means of DFT + U calculations combined with crystallographic and electrostatic analyses. We show that the triplite polymorph, although characterized by two distinct edge-shared crystallographic sites, locally exhibits corner-sharing connections between consecutive FeO4F2 octahedra, exactly as in the tavorite polymorph. As a consequence, edge-sharing connections exist in the lithiated triplite structure between consecutive FeO4F2 and LiO4F2 polyhedra. We then demonstrate that the origin of the voltage increase lies in the difference in the anionic networks of the two polymorphs, and more specifically in the electrostatic repulsions induced by the configuration of the fluorine atoms around the transition metal in the two polymorphs (trans- vs. cis-configurations in tavorite vs. triplite polymorphs). Such a finding should help in the design of novel high potential fluorosulphate materials, which beyond enhanced performances present sustainability attributes as they can be made from abundant elements and via low temperature eco-efficient processes.

 Please wait while we load your content...
Please wait while we load your content...