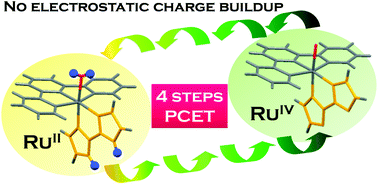
Two new ruthenium(II) complexes bearing dissociable protons, [Ru(trpy)(H2bim)Cl]PF6 (1) and [Ru(trpy)(H2bim)(OH2)](PF6)2 (2) (H2bim = 2,2′-biimidazole and trpy = 2,2′:6′,2′′-terpyridine), were synthesized and characterized, where the H2bim and M–OH2 moieties are expected to serve as proton-dissociation sites. Single crystal X-ray diffraction analyses revealed that the H2bim and M–OH2 moieties act as proton donors in intermolecular hydrogen bonds. Two pKa values of 2 (pKa1 = 9.0 and pKa2 = 11.3) were spectrophotometrically determined, where the first proton dissociation is assigned to that from H2bim and the second is from M–OH2. This assignment was supported by the density functional theory (DFT) and time-dependent density functional theory (TD-DFT) calculations for two sets of conjugated bases, [Ru(trpy)(Hbim)(OH2)]+ and [Ru(trpy)(H2bim)(OH)]+ for the first proton dissociation, and [Ru(trpy)(Hbim)(OH)]+ and [Ru(trpy)(bim)(OH2)]+ for the second dissociation. Electrochemical studies in aqueous solutions under various pH conditions afforded the Pourbaix diagram (potential versus pH diagram) of 2, where the pKa values found from the diagram agree well with those determined spectrophotometrically. It was also found that 2 demonstrates four-step proton-coupled electron transfer (PCET) reactions to give the four-electron oxidized species, [RuIV(trpy)(bim)(O)]2+, without electrostatic charge buildup during the reactions. The multiple PCET ability of 2 would be applicable to various multi-electron oxidation reactions. Catalysis of electrochemical water oxidation was indeed evaluated in the initial attempt to demonstrate multi-electron oxidation reactions, revealing that the water oxidation potential for 2 is lower than that for other ruthenium catalysts, [Ru(trpy)(bpy)(OH2)]2+, [Ru(trpy)(bpm)(OH2)]2+ and [Ru(tmtacn)(bpy)(OH2)]2+ (bpy = 2,2′-bipyridine, bpm = 2,2′-bipyrimidine, and tmtacn = 1,4,7-trimethyl-1,4,7-triazacyclononane), which are known to be active catalysts for water oxidation.

 Please wait while we load your content...
Please wait while we load your content...