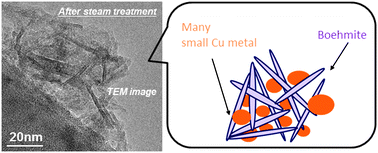
To compare the catalytic performances against daily start-up and shut-down (DSS) operations between co-precipitated (CP) and impregnated (IMP) Cu–Al–Ox catalysts for the water gas shift (WGS) reaction, in situ X-ray adsorption fine structure (XAFS) measurements, temperature-programmed reduction (TPR) profiles, X-ray diffraction (XRD) patterns and high resolution transition electron microscopy with an energy dispersive X-ray spectroscopy (TEM-EDS) analysis were performed. In situ XAFS studies clearly indicated that the Cu species were frequently oxidized and reduced during DSS operations with steam treatment (DSS-like operation). Based on in situ XAFS and H2-TPR profiles, the highly active and stable CP-catalyst possessed more susceptible Cu particles to oxidation/reduction (described as redoxable) than the IMP-catalyst even after the DSS-like operations. Interestingly, the XRD and TEM-EDS analysis showed small Cu particles which were covered with a card-house structure of the in situ formed boehmite in the case of the CP-catalyst after the DSS-like operations. According to these results, we concluded that the superior durability of the CP-catalyst against frequent redox changes was attributed to the nanoscale coordination with the in situ formed boehmite structure which preserves the small-redoxable Cu particles.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...