Photocatalytic oxidative desulfurization of dibenzothiophene under simulated sunlight irradiation with mixed-phase Fe2O3 prepared by solution combustion†
Abstract
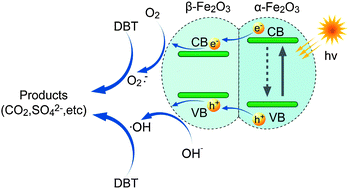
Mixed-phase Fe2O3 was prepared from Fe(NO3)3 and (C2H5)3NHCl via solution combustion. Photocatalytic oxidative desulfurization of dibenzothiophene (DBT) under simulated sunlight irradiation using mixed-phases of α- and β-Fe2O3 as catalysts was investigated. The DBT in the oil phase was extracted into water phase and then photooxidized to CO2 and SO42− by Fe2O3 and O2 dissolved in water. The Fe2O3 containing 36.6% β-Fe2O3 and 63.4% α-Fe2O3 exhibited the highest photocatalytic activity. Sulfur removal of DBT in n-octane was 92.3% for 90 min irradiation under the conditions of V(water) : V(n-octane) = 1 : 1, air flow rate at 150 mL min−1, and Fe2O3 addition at 0.05 g. The kinetics of photooxidative desulfurization of DBT was a pseudo-first-order with an apparent rate constant of 0.0287 min−1 and half-time of 24.15 min. The sulfur content of the actual diesel could be reduced from 478 μg mL−1 to 44.5 μg mL−1 after 90 min. The radical scavengers experiments and terephthalic acid fluorescence technique indicated that ˙OH and O2˙− were the main reactive species in the DBT photocatalytic degradation. A potential photocatalytic desulfurization mechanism using mixed-phase Fe2O3 was proposed.

 Please wait while we load your content...
Please wait while we load your content...