Self-disproportionation of enantiomers via achiral chromatography: a warning and an extra dimension in optical purifications
Abstract
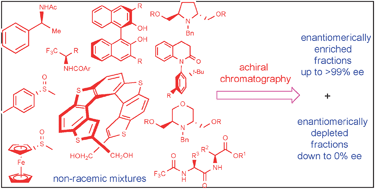
This tutorial review describes the self-disproportionation of enantiomers (SDE) of chiral, non-racemic compounds, subjected to chromatography on an achiral stationary phase using an achiral eluent, which leads to the substantial enantiomeric enrichment and the corresponding depletion in different fractions, as compared to the enantiomeric composition of the starting material. The physicochemical background of SDE is a dynamic formation of homo- or heterochiral dimeric or oligomeric aggregates of different chromatographic behavior. This phenomenon is of a very general nature as the SDE has been reported for different classes of organic compounds bearing various functional groups and possessing diverse elements of chirality (central, axial and helical chirality). The literature data discussed in this review clearly suggest that SDE via achiral chromatography might be expected for any given chiral enantiomerically enriched compound. This presents two very important issues for organic chemists. First, chromatographic purification of reaction products can lead to erroneous determination of the stereochemical outcome of catalytic asymmetric reactions and second, achiral chromatography can be used as a new, nonconventional method for optical purifications. The latter has tremendous practical potential as the currently available techniques are limited to crystallization or chiral chromatography. However, a further systematic study of SDE is needed to develop understanding of this phenomenon and to design practical chromatographic separation techniques for optical purification of non-racemic mixtures by achiral-phase chromatography.

 Please wait while we load your content...
Please wait while we load your content...