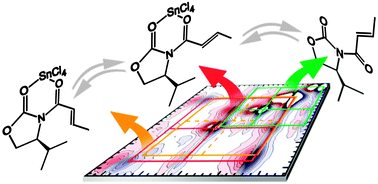
The understanding of reaction mechanisms requires structure elucidation of short-lived intermediates, even in the presence of other, similar structures. Here we show that polarization dependent two-dimensional infrared spectroscopy is a powerful method to determine the structure of molecules that participate in fast equilibria, in a regime where standard techniques such as nuclear magnetic resonance spectroscopy are beyond their limits. Using catalyst–substrate complexes in a Lewis acid catalyzed enantioselective Diels–Alder reaction as an example we present two methods that allow the resolution of molecular structure in mixtures even when the spectroscopic signals partially overlap. The structures of N-crotonyloxazolidin-2-one, a reactant carrying the Evans auxiliary, and its complex with the Lewis acid SnCl4 were determined in a mixture as used under the typical reaction conditions. In addition to the chelate that mainly forms, three additional substrate–catalyst complexes were detected and could be tentatively assigned. Observation of minor complex conformers suggests a rationale for the observed diastereoselectivity of the reaction using SnCl4 as compared to other Lewis acids. Knowledge about additional species may lead to a better understanding of the different selectivities for various Lewis acids and allow reaction optimization.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...