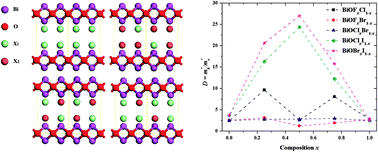
Via density functional theory computations, we investigated the photocatalytic activities of pure and alloyed bismuth oxyhalides (BiOXs). The dipole moments of the majority of pure and alloyed BiOXs are larger than 2.00 Debye, which can ensure their high photocatalytic efficiencies. Both the redox potentials of the photon-induced holes and the band gaps increase with an increasing content of lighter halogen atoms in the BiOXs, which competitively affects the photocatalytic efficiency. The hole mobility decreases a lot due to the hybridization of the halogennp states, while the electron mobility is not affected by such hybridization. Therefore, the alloying effect in BiOXs brings about a substantially lower electron–hole recombination rate and, accordingly, a much higher photocatalytic efficiency. Our investigation also suggests that O vacancies, which are energetically more favorable in alloyed BiOXs, could act as capture centers for excited electrons and, consequently, improve the separation of the electron–hole pairs. Our findings present a reasonable explanation for the recent experimental reports and provide some guidance for the design of alloyed BiOX photocatalysts.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...