The RNA core weakly influences the interactions of the bacteriophage MS2 at key environmental interfaces
Abstract
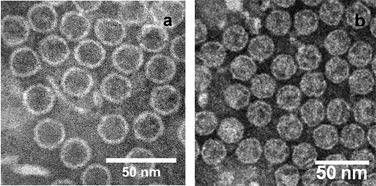
The effect of the RNA core on interfacial interactions of the bacteriophage MS2 was investigated. After removal of the RNA core, empty intact capsids were characterized and compared to untreated MS2. Electron density of untreated MS2 and RNA-free

 Please wait while we load your content...
Please wait while we load your content...