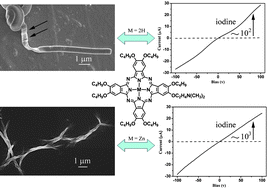
The self-assembling properties of metal free unsymmetrical 2,3,9,10,16,17,23-heptakis(butyloxy)-24-mono(dimethylaminoethyloxy)phthalocyanine H2{Pc(OC4H9)7[OC2H4N(CH3)2]} (1) and its zinc complex counterpart Zn{Pc(OC4H9)7[OC2H4N(CH3)2]} (2) in MeOH have been comparatively investigated by scanning electron microscopy (SEM), X-ray diffraction (XRD) technique, and IR and UV-vis spectroscopy. Cooperation of the intermolecular hydrogen bonding/Zn–N coordination bonding with π–π interaction between neighboring phthalocyanine molecules due to the presence of one peripheral dimethylaminoethyloxy side chain for 1 and 2 results in nanostructures with different morphology including left- and right-handed helical ribbons, as well as hollow nanotubes for 1 and nanowire bundles for 2. Electronic absorption spectroscopic results reveal the H-aggregate nature in all the nanostructures formed from both compounds 1 and 2, indicating the dominant π–π inter-molecular interaction between phthalocyanine molecules. The IR and X-ray diffraction (XRD) results reveal a dimeric supramolecular structure formed through the intermolecular hydrogen bonding or Zn–N coordination bonding interaction between two molecules, which as the building block further packs into the target nanostructures depending on π–π interaction with the help of hydrogen bonding/Zn–N coordination bonding interaction between the dimeric supramolecular building blocks for 1 and 2. In particular, the morphological evolution of the nanostructures from helical ribbons to nanotubes has been clearly revealed during the self-assembly process of unsymmetrical phthalocyanine compound 1. In addition, the nanostructures self-assembled from both 1 and 2 were revealed to show good semiconducting properties with the conductivity in the range of 2.04 × 10−5 to 1.28 × 10−4 S m−1 in air due to the ordered one-dimensional π–π stacking enhanced by hydrogen bonding/metal–ligand coordination bonding interaction, which increases to 2.41 × 10−2–2.74 × 10−2 S m−1 after chemical doping with iodine.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...