Combinatorial synthesis of metal–organic frameworks libraries by click-chemistry†
Abstract
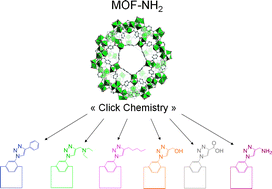
A key to address advanced MOF materials suitable for more sophisticated applications is to add functionalities of greater complexity in a controlled manner. We report a generic and original post-synthetic modification method starting from

 Please wait while we load your content...
Please wait while we load your content...