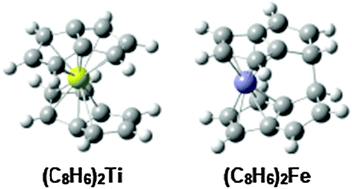
Neutral bis(pentalene)metal sandwich compounds have been synthesized for the first row transition metals titanium and iron. In this connection, the complete series of first row transition metal bis(pentalene)metal complexes (C8H6)2M (M = Ti, V, Cr, Mn, Fe, Co, Ni) has now been investigated by density functional theory in order to evaluate the effect of the metal electronic requirements on the hapticity of the pentalene ligands. The lowest energy structure for the titanium complex is the 18-electron complex (η8-C8H6)(η6-C8H6)Ti with one octahapto and one hexahapto pentalene ligand rather than the previously suggested 20-electron complex (η8-C8H6)2Ti with two octahapto pentalene ligands. The experimental NMR observations on (C8H6)2Ti can then be reinterpreted as interchange of octahapto and hexahapto bonding of the pentalene units through a low energy bis(octahapto) (η8-C8H6)2Ti transition state. The lowest energy structure for (C8H6)2V has two fulvene-like hexahapto pentalene ligands and a local vanadium environment similar to the well-known dibenzenevanadium. The lowest energy structures of the later first row transition metals have two pentahapto pentalene ligands with local metal environments similar to those in the corresponding metallocenes. In the manganese and iron structures of this type, the remaining unpaired electrons on one of the uncomplexed carbon atoms in each η5-C8H6 system form a C–C single bond to couple the two pentalene ligands. This coupling of the two pentalene systems through a C–C single bond is similar to that found experimentally by X-ray crystallography in bis(pentalene)iron, (η5-C8H6)2Fe.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...