Formation and photocatalytic properties of bismuth ferrite submicrocrystals with tunable morphologies
Abstract
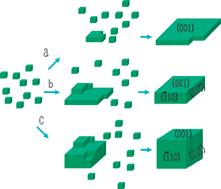
We report the synthesis of ternary bismuth ferrites Bi2Fe4O9 submicrocrystals with tunable morphologies from sheets, plates, to cubes under hydrothermal conditions. These Bi2Fe4O9 submicrocrystals were single crystal with dominating {001}, {110} and {![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) 10} facets. The crystal growth and the influence of hydroxide on the shape formation and morphology control of the Bi2Fe4O9 submicrocrystals are explored. Anisotropic growth on the (001) facets has been achieved by increasing the concentration of hydroxide, and an oriented attachment mechanism has been suggested for the formation of the Bi2Fe4O9 submicrocrystals. The photocatalytic activities of the Bi2Fe4O9 submicrocrystals have also been investigated. The results show that Bi2Fe4O9 submicrocrystals have promise as a novel material for photocatalytic applications.
10} facets. The crystal growth and the influence of hydroxide on the shape formation and morphology control of the Bi2Fe4O9 submicrocrystals are explored. Anisotropic growth on the (001) facets has been achieved by increasing the concentration of hydroxide, and an oriented attachment mechanism has been suggested for the formation of the Bi2Fe4O9 submicrocrystals. The photocatalytic activities of the Bi2Fe4O9 submicrocrystals have also been investigated. The results show that Bi2Fe4O9 submicrocrystals have promise as a novel material for photocatalytic applications.

 Please wait while we load your content...
Please wait while we load your content...