Porous nano-MnO2: large scale synthesis via a facile quick-redox procedure and application in a supercapacitor
Abstract
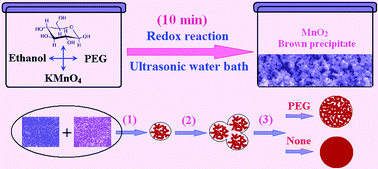
A new type of porous nano-MnO2 for supercapacitors has been synthesized for the first time by a facile sonochemistry route from a quick-

 Please wait while we load your content...
Please wait while we load your content...