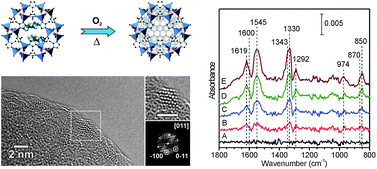
The microporous and activated zeolitic imidazolate framework (Zn(MeIM)2; MeIM = imidazolate-2-methyl; ZIF-8) was loaded with the MOCVD precursor diethyl zinc [Zn(C2H5)2]. Exposure of ZIF-8 to the vapour of the volatile organometallic molecule resulted in the formation of the inclusion compound [Zn(C2H5)2]0.38@ZIF-8 revealing two precursor molecules per cavity. In a second step the obtained material was treated with oxygen (5 vol% in argon) at various temperatures (oxidative annealing) to achieve the composite material ZnO0.35@ZIF-8. The new material was characterized with powder XRD, FT-IR, UV-vis, solid state NMR, elemental analysis, N2 sorption measurements, and transmission electron microscopy. The data give evidence for the presence of nano-sized ZnO particles stabilized by ZIF-8 showing a blue-shift of the UV-vis absorption caused by quantum size effect (QSE). The surface structure and reactivity of embedded ZnO nanoparticles were characterized viacarbon dioxide adsorption at different temperatures monitored by ultra-high vacuum FTIR techniques. It was found that the surface of ZnO nanoparticles is dominated by polar O–ZnO and Zn–ZnO facets as well as by defect sites, which all exhibit high reactivity towards CO2 activation forming various adsorbed carbonate and chemisorbed CO2δ− species.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...