Synthesis, crystallization, electrochemistry and single crystal X-ray analysis of a methoxy-substituted-tris-phenalenyl based neutral radical†‡
Abstract
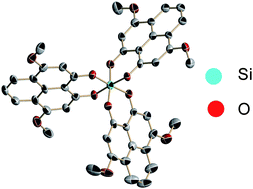
We report the synthesis, crystallization, electrochemistry, X-ray structure and electron density distribution of a methoxy-substituted-tris-phenalenyl based neutral radical (18); the cyclic voltammogram of the compound shows that the strategy of oxygen substitution at 3,7-positions and the utilization of tris-phenalenyls within the silicon complex reduce the electrochemical disproportionation potentials. The crystal structure shows that the radical exists as a 1D π-chain of partially superimposed phenalenyl rings with intermolecular carbon⋯carbon contacts that fall within van der Waals atomic separation. A bond length–bond order regression analysis of the experimental structures shows that the electron density is distributed over two out of the three phenalenyl units.
- This article is part of the themed collection: Celebrating the 70th birthday of Professor Fred Wudl

 Please wait while we load your content...
Please wait while we load your content...