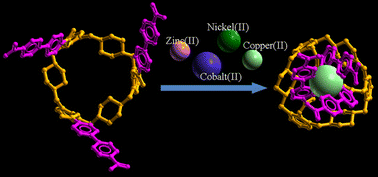
New tripodal transition metal ion receptors, tris(5-ethoxycarbonyl-2,2′-bipyridine) and tris(5-carboxylate-2,2′-bipyridine) substituted 27-membered trimeric piperazine cyclophanes 5 and 7 as well as tetra(5-ethoxycarbonyl-2,2′-bipyridine) substituted 36-membered tetrameric piperazine cyclophane 6, have been prepared and their transition metal ion complexing properties studied in solution by UV-vis spectroscopy and in the solid state by single-crystal X-ray diffraction. The crystal structures of [H353+·Fe2+]·4(ClO4−)·CF3COO− (V), [H372+·Fe2+]·2(SO42−) (VII) and the reference complex [tris(5,5′-bis(ethoxycarbonyl)-2,2′-bipyridine)Fe(II) perchlorate] (I) showed that the robust piperazine cyclophane is an optimal platform in preorganizing the 2,2′-bipy moieties to form a very fixed octahedral coordination site. In an acidic water solution, the highly preorganized structure of 5 gives a [5·Fe2+] complex, the stability of which is comparable with the classical tris(2,2′-bipy) Fe2+-complex but it is a significant 3.7 logK units more stable than the non-preorganized tetrameric analog [6·Fe2+]. Detailed studies with other similar divalent octahedral transition metal cations showed that the restricted octahedral coordination in complexes of 5 results in an unusual selectivity. The selectivity order [Zn2+<Co2+<Ni2+≈Fe2+<Cu2+] deviates significantly from that of typical tris(2,2′-bipy) complexes [Zn2+<Co2+<Cu2+≈ Fe2+<Ni2+]. As a highlight, cyclophane 5 exhibits exceptionally high selectivity towards Cu2+ ions but very weak binding for Co2+.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...