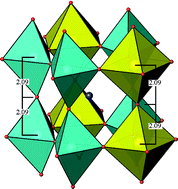
The high-pressure behaviour of (NH4)2V3O8 with the fresnoite structure (P4bm, Z = 2) has been studied at room temperature with single-crystal X-ray diffraction in diamond anvil cells using laboratory and synchrotron facilities. At ambient conditions, the crystal structure is composed of layers of corner-sharing V5+O4 tetrahedra and V4+O5 square pyramids separated by layers of the NH4+ cations. At about 3 GPa, there occurs a reversible first-order phase transition to a three-dimensional structure (P4/mbm, Z = 2) built of corner-sharing V5+O5 trigonal bipyramids and V4+O6 octahedra. The NH4+ cations fill up the interstitial sites in the tunnels formed by the vanadate framework. Up to the phase transition, the a lattice parameter of the low-pressure polymorph does not change while the contraction perpendicular to the stacking of the V3O8 slabs accounts entirely for the bulk compressibility. Above the phase transition, the a lattice parameter slightly expands. The structural features of the high-pressure phase of (NH4)2V3O8 are compared to those of other vanadium oxides.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...