The radical mechanism of cobalt(ii) porphyrin-catalyzed olefin aziridination and the importance of cooperative H-bonding†
Abstract
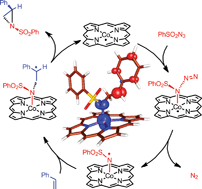
The mechanism of cobalt(II) porphyrin-mediated aziridination of ![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C double bond of
C double bond of ![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O and N–H units. The computed radical-type mechanism agrees well with all available mechanistic and kinetic information. The computed free energy profile readily explains the superior performance of the CoII(porAmide) system with H-bond donor functionalities over the non-functionalized Co(TPP).
O and N–H units. The computed radical-type mechanism agrees well with all available mechanistic and kinetic information. The computed free energy profile readily explains the superior performance of the CoII(porAmide) system with H-bond donor functionalities over the non-functionalized Co(TPP).

 Please wait while we load your content...
Please wait while we load your content...