Reaction of the deprotonated N-thiophosphorylated thioureas RNHC(S)NHP(S)(OiPr)2 (R = Ph, HLI; 2-MeC6H4–, HLII; 2,6-Me2C6H3–, HLIII; 2,4,6-Me3C6H2–, HLIV; Me–, HLV) with NiII leads to complexes of the formula [NiLI–V2]. The molecular structures of the thioureasHLII–V and the complexes [NiLII–V2] in the solid were elucidated by single-crystal X-ray diffraction analysis. In the complexes, the metal is found to be in a square planar trans-N2S2 ([NiLII–IV2]) environment formed by the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) S sulfur atoms and the P–N nitrogen atoms, or in a square planar trans-S2S′2 ([NiLV2]) environment formed by the C
S sulfur atoms and the P–N nitrogen atoms, or in a square planar trans-S2S′2 ([NiLV2]) environment formed by the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) S and P
S and P![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) S sulfur atoms of two deprotonated ligands. DFT calculations confirmed that the [Ni(LII–IV-N,S)2] isomers are more stable (by 16–21 kcal mol−1) than the corresponding [Ni(LII–IV-S,S′)2] conformers. The main reason for higher stability of the 1,3-N,S vs. 1,5-S,S′ isomers is the formation of intramolecular N–H⋯S
S sulfur atoms of two deprotonated ligands. DFT calculations confirmed that the [Ni(LII–IV-N,S)2] isomers are more stable (by 16–21 kcal mol−1) than the corresponding [Ni(LII–IV-S,S′)2] conformers. The main reason for higher stability of the 1,3-N,S vs. 1,5-S,S′ isomers is the formation of intramolecular N–H⋯S![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) P hydrogen bonds. In solution the complexes [Ni(LII–V-N,S)2] have an exclusive 1,3-N,S coordination, while the compound [Ni(LI-N,S)2] exhibits two isomers in the 1H and 31P NMR spectra. The major species is assigned to the 1,3-N,S coordinated isomer, while the minor (∼25%) signals are due to the 1,5-S,S′ isomer. UV-Vis spectroscopic results are in line with this. The electrochemical measurements reveal reversible one-electron reduction and irreversible oxidations, both assigned to ligand-centred processes.
P hydrogen bonds. In solution the complexes [Ni(LII–V-N,S)2] have an exclusive 1,3-N,S coordination, while the compound [Ni(LI-N,S)2] exhibits two isomers in the 1H and 31P NMR spectra. The major species is assigned to the 1,3-N,S coordinated isomer, while the minor (∼25%) signals are due to the 1,5-S,S′ isomer. UV-Vis spectroscopic results are in line with this. The electrochemical measurements reveal reversible one-electron reduction and irreversible oxidations, both assigned to ligand-centred processes.
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) S sulfur atoms and the P–N
S sulfur atoms and the P–N ![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) S and P
S and P![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) S sulfur atoms of two deprotonated
S sulfur atoms of two deprotonated ![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) P hydrogen bonds. In solution the complexes [Ni(LII–V-N,S)2] have an exclusive 1,3-N,S coordination, while the compound [Ni(LI-N,S)2] exhibits two isomers in the 1H and
P hydrogen bonds. In solution the complexes [Ni(LII–V-N,S)2] have an exclusive 1,3-N,S coordination, while the compound [Ni(LI-N,S)2] exhibits two isomers in the 1H and 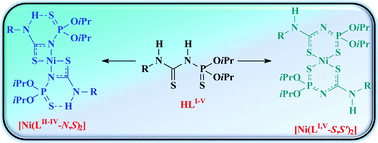

 Please wait while we load your content...
Please wait while we load your content...