Dissociative photoionization mechanism of methanol isotopologues (CH3OH, CD3OH, CH3OD and CD3OD) by iPEPICO: energetics, statistical and non-statistical kinetics and isotope effects
Abstract
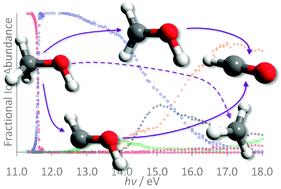
The dissociative photoionization of energy selected ![[B with combining tilde]](https://www.rsc.org/images/entities/i_char_0042_0303.gif) and
and ![[C with combining tilde]](https://www.rsc.org/images/entities/i_char_0043_0303.gif) ion states, a hydroxyl radical loss appears yielding CH3+/CD3+. Based on the branching ratios, statistical considerations and ab initio calculations, this process is confirmed to take place on the first electronically excited Ã2A′ ion state. Uncharacteristically, internal conversion is outcompeted by unimolecular dissociation due to the apparently weak Renner–Teller-like coupling between the
ion states, a hydroxyl radical loss appears yielding CH3+/CD3+. Based on the branching ratios, statistical considerations and ab initio calculations, this process is confirmed to take place on the first electronically excited Ã2A′ ion state. Uncharacteristically, internal conversion is outcompeted by unimolecular dissociation due to the apparently weak Renner–Teller-like coupling between the ![[X with combining tilde]](https://www.rsc.org/images/entities/i_char_0058_0303.gif) and the à ion states. The experimental 0 K appearance energies of the ions CH2OH+, CD2OH+, CH2OD+ and CD2OD+ are measured to be 11.646 ± 0.003 eV, 11.739 ± 0.003 eV, 11.642 ± 0.003 eV and 11.737 ± 0.003 eV, respectively. The E0(CH2OH+) = 11.6454 ± 0.0017 eV was obtained based on the independently measured isotopologue results and calculated zero point effects. The 0 K heat of formation of CH2OH+, protonated formaldehyde, was determined to be 717.7 ± 0.7 kJ mol−1. This yields a 0 K heat of formation of CH2OH of −11.1 ± 0.9 kJ mol−1 and an experimental 298 K
and the à ion states. The experimental 0 K appearance energies of the ions CH2OH+, CD2OH+, CH2OD+ and CD2OD+ are measured to be 11.646 ± 0.003 eV, 11.739 ± 0.003 eV, 11.642 ± 0.003 eV and 11.737 ± 0.003 eV, respectively. The E0(CH2OH+) = 11.6454 ± 0.0017 eV was obtained based on the independently measured isotopologue results and calculated zero point effects. The 0 K heat of formation of CH2OH+, protonated formaldehyde, was determined to be 717.7 ± 0.7 kJ mol−1. This yields a 0 K heat of formation of CH2OH of −11.1 ± 0.9 kJ mol−1 and an experimental 298 K

 Please wait while we load your content...
Please wait while we load your content...