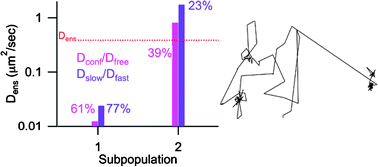
Four techniques for analyzing single molecule tracking data—confinement level analysis, time series analysis and statistical analysis of lateral diffusion, multistate kinetics, and a newly developed method, radius of gyration evolution analysis—are compared using a set of sample fluorophore trajectories obtained from the lipophilic carbocyanine dye 1,1′-dioctadecyl-3,3,3′3′-tetramethylindocarbocyanine, DiIC18, partitioned into surface tethered poly(n-isopropylacrylamide). The purpose here is two-fold: first to test that these techniques can be applied to single molecules trajectories, which typically contain a smaller total number of frames than those obtained from other particles, e.g.quantum dots or gold nanoparticles; and second to critically compare the information obtained from each method against the others. A set of five SMT trajectories, ranging in length from 41 to 273 steps with a 30 ms frame transfer exposure, were all successfully analyzed by all four techniques, provided two important criteria were met: enough steps to define the motion were acquired in the trajectory, generally on the order of 50 steps, and the fast and slow diffusion coefficients differ by at least a factor of 5. Beyond that the four trajectory analysis methods studied provide partially confirmatory and partially complementary information. SMT data resulting from more complex physical behavior may well benefit from using these techniques in succession to identify and sort populations.

 Please wait while we load your content...
Please wait while we load your content...