The fluorine atom as a halogen bond donor, viz. a positive site†
Abstract
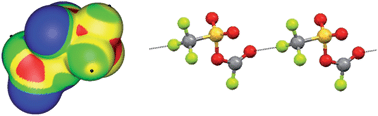
When bound to residues which can work as particularly strong electron withdrawing groups, fluorine can display a region of positive electrostatic potential (positive σ-hole). Fluorine can thus function as a

 Please wait while we load your content...
Please wait while we load your content...