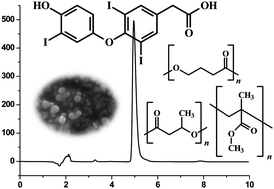
The objective of this work was to develop and validate a rapid Reversed-Phase High-Performance Liquid Chromatography method for the quantification of 3,5,3′-triiodothyroacetic acid (TRIAC) in nanoparticles delivery system prepared in different polymeric matrices. Special attention was given to developing a reliable reproductive technique for the pretreatment of the samples. Chromatographic runs were performed on an Agilent 1200 Series HPLC with a RP Phenomenex® Gemini C18 (150 × 4, 6 mm i.d., 5 μm) column using acetonitrile and triethylamine buffer 0.1% (TEA) (40 : 60 v/v) as a mobile phase in an isocratic elution, pH 5.6 at a flow rate of 1 ml min−1. TRIAC was detected at a wavelength of 220 nm. The injection volume was 20 μl and the column temperature was maintained at 35 °C. The validation characteristics included accuracy, precision, specificity, linearity, recovery, and robustness. The standard curve was found to have a linear relationship (r2 = 0.9996) over the analytical range of 5–100 μg ml−1. The detection and quantitation limits were 1.3 and 3.8 μg ml−1, respectively. The recovery and loaded TRIAC in colloidal system delivery was nearly 100% and 98%, respectively. The method was successfully applied in polycaprolactone, polyhydroxybutyrate, and polymethylmethacrylate nanoparticles.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...