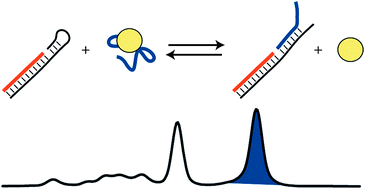
Automated microchip electrophoresis was used as a simple and rapid method to measure effective dissociation constants (Kd,eff) of aptamers against both large and small molecule targets. Human thrombin, immunoglobulin E (IgE), and adenosine triphosphate (ATP) were selected as model analytes to validate the method, with four ligands including two DNA aptamers for thrombin (two distinct epitopes), an IgE aptamer, and an ATP aptamer. The approach is based on a microchip version of a DNA mobility shift assay. Non-denaturing microchip gel electrophoresis separations of DNA could resolve and quantify unbound from target-bound aptamers when using large molecules as targets. To extend the technique to small molecule targets such as ATP, an aptamer/competitor strategy was used, in which a DNA competitor complementary to the aptamer could be displaced by ATP and electrophoretically resolved. Using an automated microchip electrophoresis platform, parallel separations of 11 titration samples were completed in ∼0.5 h. Analytical performance comparisons show that our approach provides significant advantages in minimized reagent consumption (typically tens of pmol of aptamer and target), reduced analysis time, and minimized user interaction when compared to previously reported methods for aptamerKd measurement. Moreover, the flexibility and ease of Kd,eff measurement for aptamers against large and small targets make this a unique and valuable approach that should find widespread use. Finally, the feasibility of using this method during aptamer selection processes (e.g. SELEX) was shown by accurate bulk Kd,eff measurement of a known thrombin aptamer (THRaptA) spiked into a random-sequence DNA pool at as low as 5.0% (molar %) of the total pool; only ∼825 fmol of total binding sequences were needed for an 11-point titration curve.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...