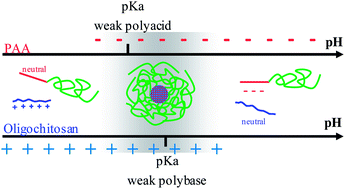
This paper reports on the polyion complex micelles (PIC micelles) formed between neutral-ionizable double hydrophilic block copolymers (DHBC), poly(ethylene oxide)-block-poly(acrylic acid) (PEO-b-PAA), and oligochitosan, a natural polyamine. The controlled synthesis of PEO-b-PAA polymers was achieved by atom transfer radical polymerization (ATRP) of tert-butyl acrylate with ω-bromide-functionalized PEO macroinitiators (Mw = 2000 and 5000 g mol−1) and the subsequent deprotection reaction under acidic conditions. A series of copolymers with a narrow molecular weight distribution (Mw/Mn ≤ 1.2) and varied PAA block lengths was synthesized. Capillary electrophoresis (CE) was shown to unambiguously prove the blocky structure of the copolymers. It also showed that about 60% of the sodium counter ions were condensed onto the polyacrylate block in the pure diblock copolymer solution, which is consistent with the formation of polyion complex micelles triggered by counter-ion release in the presence of oligochitosan. The formation of oligochitosan/PAA–PEOcore–corona micelles has been investigated by dynamic light scattering (DLS), small angle X-ray scattering (SAXS) and transmission electron microscopy (TEM). A minimum length of the PAA block is necessary to ensure micelle formation. The range of pH, where PIC micelles form, critically depends on the PAA block length, which also determines the size of the micelles. Micelles can be dissociated at ionic strength above 0.4 mol L−1. Since these PIC micelles have been used as recyclable structuring agents for the formation of ordered mesoporous materials, the reversibilty of the assembling process was studied upon pH and ionic strength cyclic variations. A hysteresis of stability was observed at low pH, probably due to hydrogen bonding.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...