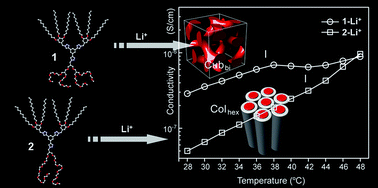
We prepared two Janus dendrimers 1 and 2 bearing a hydrophobic Percec-type minidendron (i.e., 3,4,5-tridodecyloxybenzyl group) and hydrophilic aliphatic ether dendrons with different degrees of branching via stepwise click reactions. The obtained Janus dendrimers have the same hydrophilic volume fraction (f = 0.34), but a different number of hydrophilic chains, i.e., tetrabranched and dibranched oligo(ethylene oxide) coils for 1 and 2, respectively. Upon doping with lithium salt, liquid crystalline (LC) ionic samples 1-Li+++ and 2-Li++ were obtained. As characterized by polarized optical microscopy (POM) and X-ray scattering techniques, 1-Li+++ showed an optically isotropic cubic LC phase, while 2-Li++ displayed a birefringent hexagonal columnar LC structure with a bilayered cross-section. Interestingly, the conductivity values of the cubic phase were considerably higher than those of the columnar phase, with which the cubic phase could be assigned as a bicontinuous cubic structure rather than a micellar one. On the basis of the experimental observations, the morphological contrast between two ionic Janus dendrimers is strongly attributed to the degree of chain branching, which is one of the major parameters governing the morphological features in the Janus dendrimer assemblies.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...