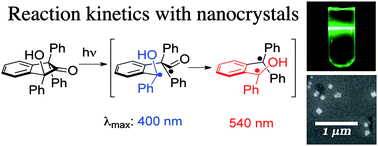
Electronic excitation of crystalline 1,3,3-triphenyl-1-hydroxy-2-indanone results in the exclusive formation of hydroxy-benzocyclobutane while irradiation in solution leads to formation of the isomeric photoenols, which subsequently tautomerize. Using nanocrystals suspended in water we were able to use transmission pump–probe methods to detect the reactive intermediates involved in the solid state. Transients assigned to the 1,4-biradical (triplet enol) formed by adiabatic photodecarbonylation were detected. It was found that product formation occurs from analogous transients, both in solution and in the solid state. While enols revert to ketones in time scales that range from a few hundred nanoseconds to tens of microseconds, benzocyclobutanol remains kinetically trapped in the crystal lattice, but undergoes a thermal ring opening when dissolved.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?

 Please wait while we load your content...
Please wait while we load your content...