The synthesis of [(1,2,4-tBu3C5H2)FeI]2, [Cp′FeI]2 (1), and its reaction chemistry are described. Complex 1 shows remarkable stability toward ligand redistribution in solution and gas-phase due to kinetic stabilization with respect to 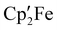 formation. In coordinating solvents the dimer is homolytically cleaved to form paramagnetic mono-solvate 16 valence-electron (VE) adducts [Cp′FeI(L)]. These solvent adducts are labile, and 1 is re-established on exposure to dynamic vacuum. Complex 1 was demonstrated to be a suitable synthon for [Cp′Fe]+ transfer to Ir pincer complexes and successful η6-coordination of this fragment to the arene ring of κ1-metallated POCOP–iridium pincer complexes [(Cp′Fe)(POCOP)Ir(H)(I)] ([5]++) and [(Cp′Fe)(POCOP)Ir(C2H4)] ([7]++) (POCOP = C6H3-2,6-(OPtBu2)2) was accomplished. In these heterometallic organometallic complexes, the π- and σ-electrons of the POCOP-pincer phenyl anion are involved in η6- and κ1-coordination to iron(II) and iridium(III) and iridium(I), respectively. [Cp′Fe]+ coordination alters the charge distribution in the (POCOP)Ir complex by inducing significant charge transfer from the POCOP aryl π-system to the [Cp′Fe]+ fragment, and therefore yielding an electron-poor and more electrophilic iridium center. Density functional theory (DFT) calculations provide a detailed understanding of this effect. Preliminary catalytic studies show that [7]++ is an active catalyst in transfer dehydrogenation between cyclooctane (COA) and tert-butyl ethylene (TBE).
formation. In coordinating solvents the dimer is homolytically cleaved to form paramagnetic mono-solvate 16 valence-electron (VE) adducts [Cp′FeI(L)]. These solvent adducts are labile, and 1 is re-established on exposure to dynamic vacuum. Complex 1 was demonstrated to be a suitable synthon for [Cp′Fe]+ transfer to Ir pincer complexes and successful η6-coordination of this fragment to the arene ring of κ1-metallated POCOP–iridium pincer complexes [(Cp′Fe)(POCOP)Ir(H)(I)] ([5]++) and [(Cp′Fe)(POCOP)Ir(C2H4)] ([7]++) (POCOP = C6H3-2,6-(OPtBu2)2) was accomplished. In these heterometallic organometallic complexes, the π- and σ-electrons of the POCOP-pincer phenyl anion are involved in η6- and κ1-coordination to iron(II) and iridium(III) and iridium(I), respectively. [Cp′Fe]+ coordination alters the charge distribution in the (POCOP)Ir complex by inducing significant charge transfer from the POCOP aryl π-system to the [Cp′Fe]+ fragment, and therefore yielding an electron-poor and more electrophilic iridium center. Density functional theory (DFT) calculations provide a detailed understanding of this effect. Preliminary catalytic studies show that [7]++ is an active catalyst in transfer dehydrogenation between cyclooctane (COA) and tert-butyl ethylene (TBE).
![Graphical abstract: [Cp′FeI]2 as convenient entry into iron-modified pincer complexes: bimetallic η6,κ1-POCOP-pincer iron iridium compounds](/en/Image/Get?imageInfo.ImageType=GA&imageInfo.ImageIdentifier.ManuscriptID=C1NJ20399A&imageInfo.ImageIdentifier.Year=2011)
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?
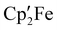 formation. In coordinating
formation. In coordinating ![Graphical abstract: [Cp′FeI]2 as convenient entry into iron-modified pincer complexes: bimetallic η6,κ1-POCOP-pincer iron iridium compounds](/en/Image/Get?imageInfo.ImageType=GA&imageInfo.ImageIdentifier.ManuscriptID=C1NJ20399A&imageInfo.ImageIdentifier.Year=2011)

 Please wait while we load your content...
Please wait while we load your content...