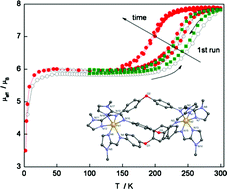
Reported herein are the synthesis, structural, magnetic and Mössbauer spectroscopic characterisation of a dinuclear Fe(II) triple helicate complex [Fe2(L)3](ClO4)4.xH2O (x = 1–4), 1(H2O), where L is a bis-bidentate imidazolimine ligand. Low temperature structural analysis (150 K) and Mössbauer spectroscopy (4.5 K) are consistent with one of the Fe(II) centres within the helicate being in the low spin (LS) state with the other being in the high-spin (HS) state resulting in a [LS:HS] species. However, Mössbauer spectroscopy (295 K) and variable temperature magnetic susceptibility measurements (4.5–300 K) reveal that 1(H2O) undergoes a reversible single step spin crossover at one Fe(II) centre at higher temperatures resulting in a [HS:HS] species. Indeed, the T1/2(SCO) values at this Fe(II) centre also vary as the degree of hydration, x, within 1(H2O) changes from 1 to 4 and are centred between ca. 210 K–265 K, respectively. The dehydration/hydration cycle is reversible and the fully hydrated phase of 1(H2O) may be recovered on exposure to water vapour. This magnetic behaviour is in contrast to that observed in the related compound [Fe2(L)3](ClO4)4·2MeCN, 1(MeCN), whereby fully reversible SCO was observed at each Fe(II) centre to give [LS:LS] species at low temperature and [HS:HS] species at higher temperatures. Reasons for this differing behaviour between 1(H2O) and 1(MeCN) are discussed.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...