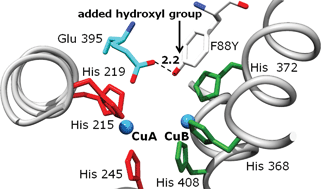
The critical review describes the known dicopper systems mediating the aromatic hydroxylation of monophenolic substrates. Such systems are of interest as structural and functional models of the type 3 copper enzyme tyrosinase, which catalyzes the ortho-hydroxylation of tyrosine to DOPA and the subsequent two-electron oxidation to dopaquinone. Small-molecule systems involving μ-η²:η² peroxo, bis-μ-oxo and trans-μ-1,2 peroxo dicopper cores are considered separately. These tyrosinase models are contrasted to copper–dioxygen systems inducing radical reactions, and the different mechanistic pathways are discussed. In addition to considering the stoichiometric conversion of phenolic substrates, the available catalytic systems are described. The second part of the review deals with tyrosinase. After an introduction on the occurrence and function of tyrosinases, several aspects of the chemical reactivity of this class of enzymes are described. The analogies between the small-molecule and the enzymatic system are considered, and the implications for the reaction pathway of tyrosinase are discussed (140 references).
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...