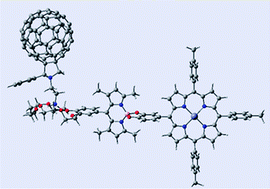
A self-assembled supramolecular triad as a model to mimic the light-induced events of the photosynthetic antenna-reaction center, that is, ultrafast excitation transfer followed by electron transfer ultimately generating a long-lived charge-separated state, has been accomplished. Boron dipyrrin (BDP), zinc porphyrin (ZnP) and fullerene (C60), respectively, constitute the energy donor, electron donor and electron acceptor segments of the antenna-reaction center imitation. Unlike in the previous models, the BDP entity was placed between the electron donor, ZnP and electron acceptor, C60 entities. For the construction, benzo-18-crown-6 functionalized BDP was synthesized and subsequently reacted with 3,4-dihydroxyphenyl functionalized ZnP through the central boron atom to form the crown-BDP-ZnP dyad. Next, an alkyl ammonium functionalized fullerene was used to self-assemble the crown ether entity of the dyad via ion–dipole interactions. The newly formed supramolecular triad was fully characterized by spectroscopic, computational and electrochemical methods. Steady-state fluorescence and excitation studies revealed the occurrence of energy transfer upon selective excitation of the BDP in the dyad. Further studies involving the pump–probe technique revealed excitation transfer from the 1BDP* to ZnP to occur in about 7 ps, much faster than that reported for other systems in this series of triads, as a consequence of shorter distance between the entities. Upon forming the supramolecular triad by self-assembling fullerene, the 1ZnP* produced by direct excitation or by energy transfer mechanism resulted in an initial electron transfer to the BDP entity. The charge recombination resulted in the population of the triplet excited state of C60, from where additional electron transfer occurred to produce C60•−:crown-BDP-ZnP•+ ion pair as the final charge-separated species. Nanosecond transient absorption studies revealed the lifetime of the charge-separated state to be ∼100 μs, the longest ever reported for this type of antenna-reaction center mimics, indicating better charge stabilization as a result of the different disposition of the entities of the supramolecular triad.

 Please wait while we load your content...
Please wait while we load your content...