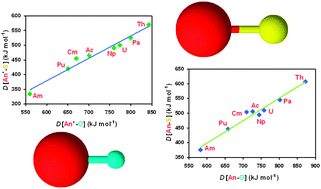
The gas-phase thermochemistry of actinide monosulfides, AnS, was investigated experimentally and theoretically. Fourier transform ion cyclotron resonance mass spectrometry was employed to study the reactivity of An+ and AnO+ (An = Th, Pa, U, Np, Pu, Am and Cm) with CS2 and COS, as well as the reactivity of the produced AnS+ with oxidants (COS, CO2, CH2O and NO). From these experiments, An+–S bond dissociation energies could be bracketed. Density functional theory studies of the energetics of neutral and monocationic AnS (An = Ac, Th, Pa, U, Np, Pu, Am and Cm) provided values for bond dissociation energies and ionization energies; the computed energetics of neutral and monocationic AnO were also obtained for comparison. The theoretical data, together with comparisons with known An+–O bond dissociation energies and M+–S and M+–O dissociation energies for the early transition metals, allowed for the refining of the An+–S bond dissociation energy ranges obtained from experiment. Examination of the reactivity of AnS+ with dienes, coupled to comparisons with reactivities of the AnO+ analogues, systematic considerations and the theoretical results, allowed for the estimation of the ionization energies of the AnS; the bond dissociation energies of neutral AnS were consequently derived. Estimates for the case of AcS were also made, based on correlations of the data for the other An and the electronic energetics of neutral and ionic An. The nature of the bonding in the elementary molecular actinide chalcogenides (oxides and sulfides) is discussed, based on both the experimental data and the computed electronic structures. DFT calculations of ionization energies for the actinide atoms and the diatomic sulfides and oxides are relatively reliable, but the calculation of bond dissociation energies is not uniformly satisfactory, either with DFT or CCSD(T). A key conclusion from both the experimental and theoretical results is that the 5f electrons do not substantially participate in actinide–sulfur bonding. We emphasize that actinides form strikingly strong bonds with both oxygen and sulfur.

 Please wait while we load your content...
Please wait while we load your content...