Temperature-cycle single-molecule FRET microscopy on polyprolines
Abstract
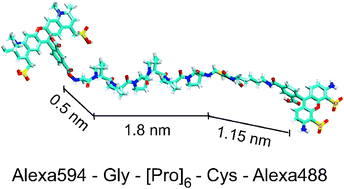
Accessing the microsecond dynamics of a single fluorescent molecule in real time is difficult because molecular fluorescence rates usually limit the time resolution to milliseconds. We propose to apply single-molecule temperature-cycle
- This article is part of the themed collection: Single-molecule optical studies of soft and complex matter

 Please wait while we load your content...
Please wait while we load your content...