Experimental and computational study of the ring opening of tricyclic oxanorbornenes to polyhydro isoindole phosphonates†‡
Abstract
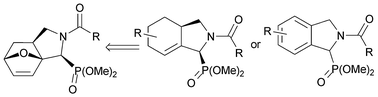
Phosphonylated azaheterocycles are an important class of compounds with high biological potential as conformationally restricted bioisosteres of amino acids. Therefore, it is of interest to synthesize conformationally constrained amino phosphonates. We wanted to investigate possible routes via ring opening of α-amino phosphonates with an

 Please wait while we load your content...
Please wait while we load your content...