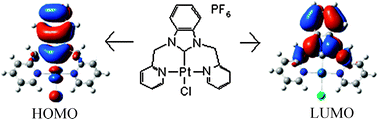
Novel NCN-pincer carbene complexes of Ru(II), 1,3-bis(2-pyridylmethyl)benzimidazolineruthenium(II) bishexafluorophosphate (2), and Pt(II), 1,3-bis(2-pyridylmethyl)benzimidazolinechloroplatinum(II) hexafluorophosphate (3), complexes based on 1,3-bis(2-pyridylmethyl)-1H-benzimidazolium chloride (1) were synthesized and characterized by different spectroscopic methods. Complex 2 shows an absorption maximum at 386 nm, blue-shifted in comparison to Ru(bpy)32+ and Ru(tpy)22+, probably due to the strong σ-donor and weak π-acceptor properties of the electron-rich NHC ligand. Electrochemical studies show Ru(II)/Ru(III) and Pt(II)/Pt(IV) reversible couples at 0.67 and 0.58 eV, respectively, lower than those for the analogous complexes of ligands like bipyridine (bpy), terpyridine (tpy) and phenylbipyridine (pbpy). The solid state structure of 2 was solved by X-ray diffraction. Theoretical studies (B3LYP/LANL2DZ) of the complex show a HOMO (−0.38594 au) mainly centered on the ruthenium and benzimidazole, whereas the LUMO (−0.25130 au) is populated by pyridines. Therefore, it is assumed that the charge transfer from HOMO → LUMO is mixed ILCT (interligand charge transfer)/MLCT (metal to ligand charge transfer). The observed lower redox potentials of the Pt(II) complex compared to the Ru(II) complex is supported by theoretically calculated ionisation potentials and also electron affinity values. To the best of our knowledge, 2 is the first example of a six-membered metallacycle homoleptic chelate pincer NCN–Ru(II) N-heterocyclic carbene complex.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...