Synthesis, characterization and biological evaluation of In(iii) complexes anchored by DOTA-like chelators bearing a quinazoline moiety†
Abstract
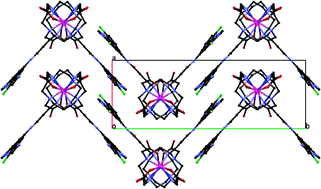
Following previous studies with a
* Corresponding authors
a Unidade de Ciências Químicas e Radiofarmacêuticas, Instituto Tecnológico e Nuclear, Estrada Nacional 10, Apartado 21, 2686-953 Sacavém, Portugal
b Centre de Biophysique Moléculaire CNRS, Rue Charles Sadron 45071 Orléans Cedex 2, France
Following previous studies with a

 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?
 Fetching data from CrossRef.
Fetching data from CrossRef.
This may take some time to load.
Loading related content
