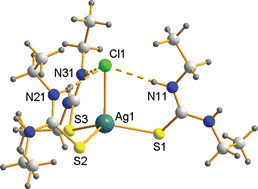
Syntheses, single crystal X-ray structural and spectroscopic characterizations are described for a variety of adducts of silver halides with thiourea (‘tu’), N-ethylthiourea (‘ettu’ = EtNH·CS·NH2) and N,N′-diethylthiourea (‘detu’ = EtNH·CS·EtNH). This study greatly extends our knowledge of the complex chemistry of silver(I) halides with tu. The unexpected diversity of composition of such complexes was initially revealed by the powerful mechanochemical/IR method and the structures of several of the new complexes were determined by X-ray crystallographic studies of samples subsequently prepared from aqueous solution. Structural characterizations provide extensive geometrical data for (mononuclear 1 : 3) [XAg(xtu)3] arrays (xtu = ettu, detu; X = Cl, Br, I), assisting vibrational spectroscopic assignments. With the parent tu ligand, binuclear ionic forms are obtained: [tu2Ag(μ-tu)2Agtu2]X2, the chloride as the dihydrate and the bromide as a di(thiourea) dihydrate adduct. With tu 1 : 2 complexes are defined for X = Cl, Br, as isomorphous one-dimensional polymers; AgCl:tu (1 : 1) is a two-dimensional net with six-membered Ag3S3 rings and terminal chloride. Intermediate stoichiometries are defined in AgBr:tu (2 : 3), a one-dimensional polymer of Ag2S2 rhombs linked at the silver atoms, while [Cl2Ag3(tu)4](NO3)·H2O is also a one-dimensional polymer in which chloride and thiourea sulfur atoms link six-membered Ag3S3 rings. Diagnostic bands in the IR and far-IR spectra of all of the synthesized complexes are assigned and interpreted in relation to the structures.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...