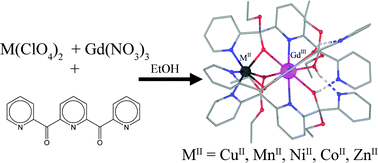
Complexes [MIIGdIII{pyCO(OEt)pyC(OH)(OEt)py}3](ClO4)2·EtOH [MII = CuII (1), MnII (2), NiII (3), CoII (4) and ZnII (5)] crystallize in the monoclinic Cc space group and contain one hexacoordinate MII ion and one enneacoordinate GdIII ion, bridged by three {pyCO(OEt)pyC(OH)(OEt)py}− ligands. Magnetic susceptibility measurements indicate a ferromagnetic interaction for 1 and antiferromagnetic interactions for 2–4. Using the Ĥ = −JŜGdIIIŜMII spin Hamiltonian formalism, fits to the magnetic susceptibility data yielded J values of +0.32 cm−1 for 1, −1.7 cm−1 for 2, and −0.22 cm−1 for 3. In complex 4, the orbital contributions of CoII precluded the determination of the magnetic coupling. The complex follows the Curie–Weiss law with θ = −2.07 K (−1.44 cm−1).
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?

 Please wait while we load your content...
Please wait while we load your content...