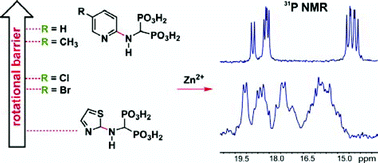
The solution properties of a series of (pyridin-2-yl)aminomethane-1,1-diphosphonic acids 1–4 as well as closely related compounds 5 and 6 with 1,3-thiazolyl or 1,3-benzothiazolyl side chains and their influence on the complexation of Zn(II), Mg(II) and Ca(II) were studied by means of the 31P and 1H NMR spectroscopy, pH-potentiometry and ESI-MS methods. The crystal structures of 5 and 6 were determined by X-ray analysis. All the studied compounds exist in solution as mixtures of the Z and E isomers with respect to the C2–Namino bond. Compounds 1 and 2 exhibit higher overall basicities compared to 3–6 and have exceptionally basic pyridyl nitrogen (pK2 = 7.88 and 8.18, respectively). Dynamic NMR studies revealed that the nature of the aromatic side chain as well as the specificity and topology of the ring substituent account for the rotational barrier for the Z/E interconversion, which decreases in the order of 1∼2 > 3∼4≫5 (6). This is particularly important for aggregational properties of 1–6 in solution and for the complex-formation equilibria. Overall, compounds 1–6 demonstrate a strong tendency for the formation of protonated multinuclear complexes. Their formation is unexpectedly slow on the NMR time scale compared to previously studied bisphosphonate complexes. The stepwise release of protons upon the rise of pH leads to mononuclear species. The Z ligand conformation is preferred in multinuclear complexes of Zn(II), Mg(II) and Ca(II) with 1 and 2 whereas in the case of 3–6 the complexation processes are affected by the intramolecular Z/E interconversion.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...