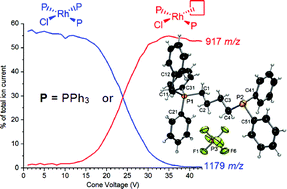
Bisphosphines Ph2P(CH2)nPPh2 (n = 1, 2, 4, 6) may be easily monobenzylated to generate cationic phosphine/phosphonium ligands [Ph2P(CH2)nPPh2CH2Ph]+. These ligands may be doped into a catalytic reaction involving neutral complexes with labile phosphine ligands, and replacement of a neutral phosphine with a charged analogue renders the resulting complex amenable to electrospray ionisation mass spectrometry (ESI-MS). Examination of olefin hydrogenation with Wilkinson's catalyst, RhCl(PPh3)3, revealed that this approach yielded rapid identification of all off-cycle solution species as well as catalyst poisons. Reactive intermediates could be generated using collision-induced dissociation (CID) of a triphenylphosphine ligand to make three-coordinate RhClP2 species, and these react with alkenes in the gas phase to form RhClP2(alkene). The solution speciation and gas phase behaviour revealed by ESI-MS match closely to what is already known about the system from kinetic and NMR studies.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?

 Please wait while we load your content...
Please wait while we load your content...