Ion-pair induced self-assembly in aqueous solvents†
Abstract
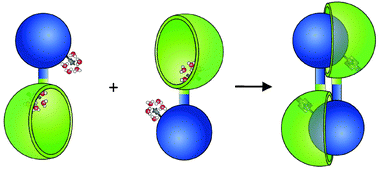
The intriguing advantages of supramolecular chemistry and particularly the application of
- This article is part of the themed collection: Supramolecular Chemistry of Anionic Species

 Please wait while we load your content...
Please wait while we load your content...