We investigate the infrared and electronic absorption spectra and the excited-state nonradiative processes of supersonic jet-cooled 5-methyl-2-hydroxypyrimidine (5M2HP), the enol form of deoxythymine, using two-color resonant two-photon ionization (R2PI) and infrared–UV depletion spectroscopies. Unlike uracil and thymine, which exhibit structureless electronic absorption spectra, the vibronic spectrum of 5M2HP is structured with narrow vibronic bands, allowing for the first time to probe the excited state of a thymine analogue. The S0 state infrared depletion spectrum shows an O–H and no N–H stretch band, identifying the spectrum as that of the enol tautomer. The S1 ← S0 electronic transition is 1nπ*, as evidenced by the rotational contour of the 000 band. Vibronic excitations of the in-plane benzene-type vibrations 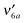 ,
, 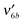 ,
, 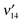 and
and 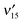 are observed, while none are observed for the out-of plane fundamental excitations, implying that the 1nπ* excited state of 5M2HP has a planar pyrimidine frame. From 1200 to 3600 cm−1 the vibronic bands become steadily broader, signaling a coupling to a lower-lying electronic state that increases with increasing energy. At ∼3600 cm−1 above the origin, the R2PI spectrum broadens completely, indicating that the two states are strongly mixed. Delayed ionization measurements show that the coupled electronic state has a >5 μs lifetime. No fluorescence has been observed from the 1nπ* state, implying relaxation to the lower-lying long-lived state is very efficient. Separate ionization potentials are measured for the 1nπ* state (9.178 eV) and for the long-lived state (∼9.46 eV), hence the latter lies ∼2200 cm−1 below the 1nπ* state. Time-dependent B3LYP calculations of the excited states of 5M2HP indeed predict the S1 state to be 1nπ* with a planar hydroxypyrimidine moiety. The T1 (3ππ*) state is calculated to lie 3000 cm−1 below the S1 state, in excellent agreement with the experiment.
are observed, while none are observed for the out-of plane fundamental excitations, implying that the 1nπ* excited state of 5M2HP has a planar pyrimidine frame. From 1200 to 3600 cm−1 the vibronic bands become steadily broader, signaling a coupling to a lower-lying electronic state that increases with increasing energy. At ∼3600 cm−1 above the origin, the R2PI spectrum broadens completely, indicating that the two states are strongly mixed. Delayed ionization measurements show that the coupled electronic state has a >5 μs lifetime. No fluorescence has been observed from the 1nπ* state, implying relaxation to the lower-lying long-lived state is very efficient. Separate ionization potentials are measured for the 1nπ* state (9.178 eV) and for the long-lived state (∼9.46 eV), hence the latter lies ∼2200 cm−1 below the 1nπ* state. Time-dependent B3LYP calculations of the excited states of 5M2HP indeed predict the S1 state to be 1nπ* with a planar hydroxypyrimidine moiety. The T1 (3ππ*) state is calculated to lie 3000 cm−1 below the S1 state, in excellent agreement with the experiment.
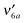 ,
, 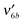 ,
, 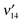 and
and 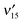 are observed, while none are observed for the out-of plane fundamental excitations, implying that the
are observed, while none are observed for the out-of plane fundamental excitations, implying that the 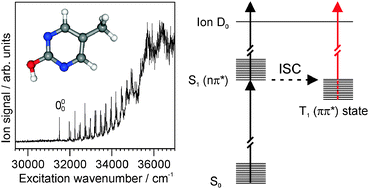

 Please wait while we load your content...
Please wait while we load your content...