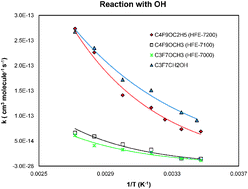
The atmospheric chemistry of several gases used in industrial applications, C4F9OC2H5 (HFE-7200), C4F9OCH3 (HFE-7100), C3F7OCH3 (HFE-7000) and C3F7CH2OH, has been studied. The discharge flow technique coupled with mass-spectrometric detection has been used to study the kinetics of their reactions with OH radicals as a function of temperature. The infrared spectra of the compounds have also been measured. The following Arrhenius expressions for the reactions were determined (in units of cm3 molecule−1 s−1): k(OH + HFE-7200) = (6.9+2.3−1.7) × 10−11 exp(−(2030 ± 190)/T); k(OH + HFE-7100) = (2.8+3.2−1.5) × 10−11 exp(−(2200 ± 490)/T); k(OH + HFE-7000) = (2.0+1.2−0.7) × 10−11 exp(−(2130 ± 290)/T); and k(OH + C3F7CH2OH) = (1.4+0.3−0.2) × 10−11 exp(−(1460 ± 120)/T). From the infrared spectra, radiative forcing efficiencies were determined and compared with earlier estimates in the literature. These were combined with the kinetic data to estimate 100-year time horizon global warming potentials relative to CO2 of 69, 337, 499 and 36 for HFE-7200, HFE-7100, HFE-7000 and CF3CF2CF2CH2OH, respectively.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...