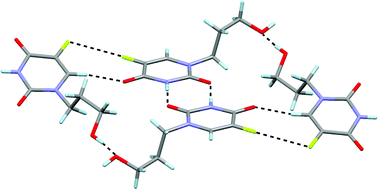
We have recently communicated the important role of lone pair–π, π–π and hydrophobic interactions in the solid architecture of 5-fluoro-1-hexyluracil and 1-hexyluracil (CrystEngComm, 2010, 12, 362–365). As a matter of fact, the simple substitution of a hydrogen atom by a fluorine atom has an enormous consequence in the solid state structure. It has been demonstrated that this is due to an increase in the π-acidity of the ring. In this article we extend the study to other uracil derivatives, where we have changed the hydrophobicity of the hexyl chain by introducing hydrophilic groups in the substituent, such as hydroxyl or carboxylic groups. The latter compounds, i.e. (N1-(3-hydroxypropyl)-5-fluorouracil and N1-(4-hydroxycarbonylbutyl)-5-fluorouracil monohydrate present interesting fluorine–fluorine interactions that are very important in determining the crystal packing.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?

 Please wait while we load your content...
Please wait while we load your content...