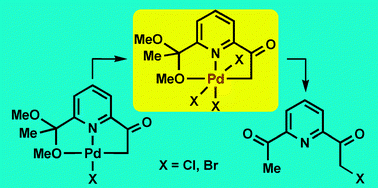
The pincer complexes [Pd(O1,N1,C1-L)X], where X = Cl, Br and L is the monoanionic ligand resulting from deprotonation of the acetyl methyl group of the monoketal of 2,6-diacetylpyridine (dap), react with excess of Cl2 or Br2 affording, quantitatively, the Pd(IV) complexes [Pd(O1,N1,C1-L)X3], which have been characterized by X-ray diffraction, and their decomposition that quantitatively affords the reductive elimination products L–X has been studied.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?

 Please wait while we load your content...
Please wait while we load your content...