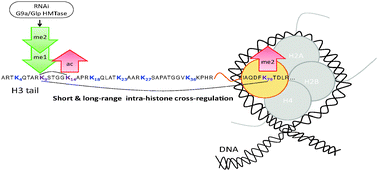
Histones are highly conserved proteins that organize cellular DNA. These proteins, especially their N-terminal domains, are adorned with many post-translational modifications (PTMs) such as lysine methylation, which are associated with active or repressed transcriptional states. The lysine methyltransferase G9a and its interaction partner Glp1 can mono- or dimethylate histone H3 on lysine (H3K9me1 or me2); possible cross-talk between these modifications and other PTMs on the same or other histone molecules is currently uncharacterized. In this study, we comprehensively analyze the effects of G9a/Glp1 knockdown on the most abundant histone modifications through both Bottom Up and Middle Down mass spectrometry-based proteomics. In addition to the expected decrease in H3K9me1/me2 we find that other degrees of methylation on K9 are affected by the reduction of G9a/Glp1 activity, particularly when K9 methylation occurs in combination with K14 acetylation. In line with this, an increase in K14 acetylation upon G9a knockdown was observed across all H3 variants (H3.1, H3.2 and H3.3), hinting at the potential existence of a binary switch between K9 methylation and K14 acetylation. Interestingly, we also detect changes in the abundance of other modifications (such as H3K79me2) in response to lowered levels of G9a/Glp1 suggesting histone PTM cross-talk amongst the H3 variants. In contrast, we find that G9a/Glp1 knockdown produces little effect on the levels of histone H4 PTMs, indicating low to no trans-histone PTM crosstalk. Lastly, we determined gene expression profiles of control and G9a/Glp1 knockdown cells, and we find that the G9a/Glp1 knockdown influences several genes, including DNA binding proteins and key factors in chromatin. Our results provide new insights into the intra- and inter- histone cross-regulation of histone K9 methylation and its potential downstream gene targets.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...