Multidimensional analysis of the frequencies and rates of cytokine secretion from single cells by quantitative microengraving†
Abstract
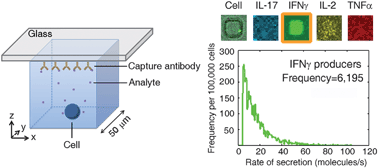
The large diversity of
* Corresponding authors
a
Department of Chemical Engineering, Massachusetts Institute of Technology, 77 Massachusetts Ave., Bldg. 66-456, Cambridge, MA, USA
E-mail:
clove@mit.edu
Fax: +617-258-5042
Tel: +617-324-2300
b Division of Molecular Immunology, Center for Neurologic Diseases, Brigham and Women's Hospital, Harvard Medical School, 77 Avenue Louis Pasteur, Boston, MA
c The Eli and Edythe L. Broad Institute, Seven Cambridge Center, Cambridge, MA, USA
d Department of Neurology, Yale University, 15 York Street, P.O. Box 208018, New Haven, CT, USA
The large diversity of

 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?
Q. Han, E. M. Bradshaw, B. Nilsson, D. A. Hafler and J. C. Love, Lab Chip, 2010, 10, 1391 DOI: 10.1039/B926849A
To request permission to reproduce material from this article, please go to the Copyright Clearance Center request page.
If you are an author contributing to an RSC publication, you do not need to request permission provided correct acknowledgement is given.
If you are the author of this article, you do not need to request permission to reproduce figures and diagrams provided correct acknowledgement is given. If you want to reproduce the whole article in a third-party publication (excluding your thesis/dissertation for which permission is not required) please go to the Copyright Clearance Center request page.
Read more about how to correctly acknowledge RSC content.
 Fetching data from CrossRef.
Fetching data from CrossRef.
This may take some time to load.
Loading related content
